Foreign Diplomats To Tour India’s Covid-19 Vaccine Facilities
Dec 9, 2020 | Pratirodh Bureau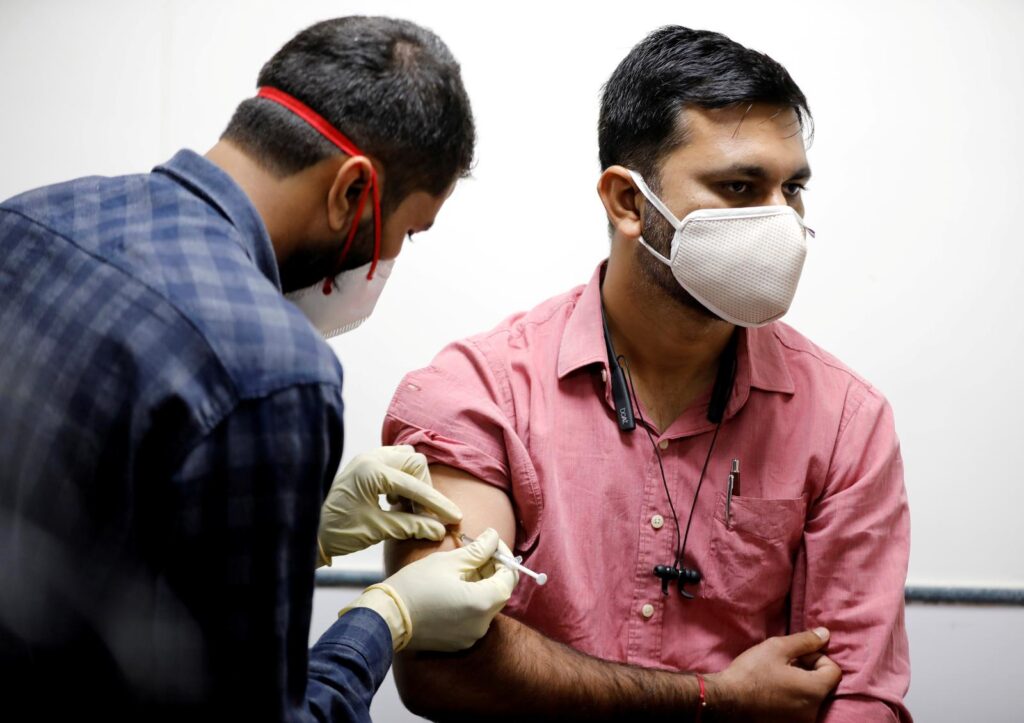
FILE PHOTO: A medic administers Covaxin to a health worker during its trials in Ahmedabad on November 26, 2020
More than 60 heads of foreign missions will visit manufacturing facilities in southern India on Wednesday, a government source said, as one of the world’s leading pharmaceutical centres steps up production of COVID-19 vaccines.
India, with the world’s second-highest tally of 9.7 million cases of the novel coronavirus, is reviewing applications from Pfizer Inc, AstraZeneca and Hyderabad-based Bharat Biotech for emergency use.
Diplomats will visit Biotech’s facilities in Hyderabad, where the firm is developing a shot called Covaxin, in collaboration with the government-run Indian Council of Medical Research, the source briefed on the plans said.
“There is a lot of interest in India’s vaccine development efforts,” the source said on Tuesday, declining to be identified because of government rules.
The visit would also include Biological E. Ltd’s facility, where the firm is developing a vaccine in collaboration with Baylor College of Medicine in Houston, Texas, and U.S.-based Dynavax Technologies Corp, the source said.
India may approve some coronavirus vaccines over the next few weeks and an estimated 300 million people would be inoculated in the first tranche, federal Health Secretary Rajesh Bhushan said on Tuesday.
Hydrabad is a major hub for pharmaceuticals in India, the world’s largest generic drug manufacturer.
